“DeltaFLU”Universal Influenza Vaccine
DeltaFLU is an advanced, self-adjuvanting influenza vaccine for protection against all influenza type A and B viruses, including seasonal influenza virus strains and emerging strains with pandemic potential. DeltaFLU is administered as a nasal spray. Immunization with DeltaFLU is simple, convenient and pain-free.
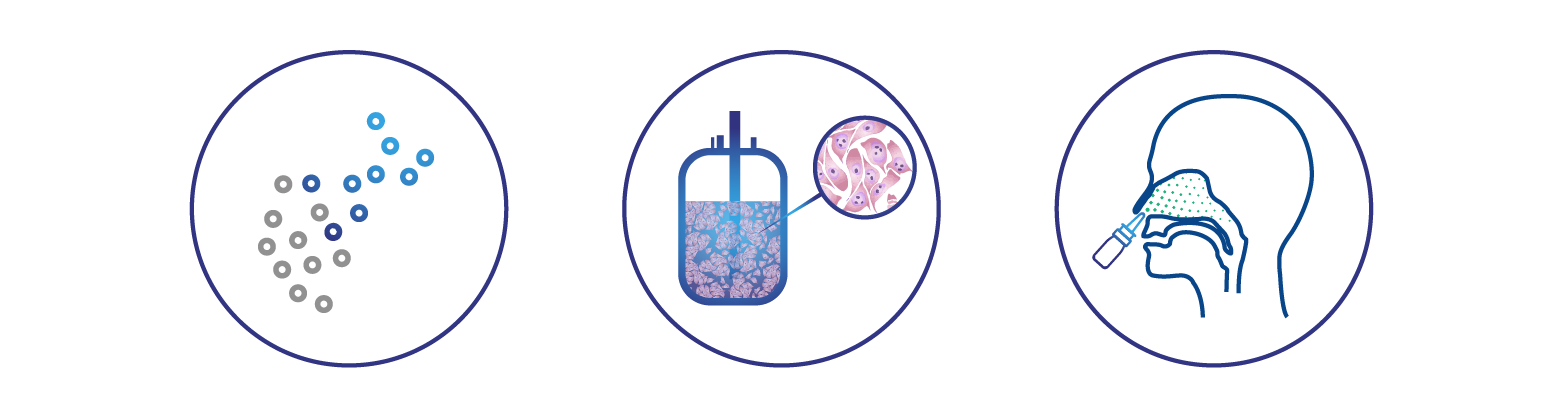
Universal protection against all influenza virus types is enabled by targeting multiple influenza virus antigens with multiple broadly protective immune mechanisms, including:
Rapid immune response with interferon and broadly protective antibodies in the nasal passages, generating a first line of defense
Antibodies targeting the conserved hemagglutinin (HA) stem
T cells targeting conserved internal antigens – systemic and tissue-resident memory T cells
We’ve already demonstrated safety and broad immune responses in clinical trials of DeltaFLU component vaccine strains. We’ve developed a Vero cell production system with proprietary technologies for high-yields, and produced vaccine strains under GMP. Our clinical trial program for universal protection starts in 2025.
The European Union is funding Phase 1 and 2 clinical studies of DeltaFLU universal influenza vaccine through FLUniversal, a consortium of Vivaldi Biosciences and leading academic and industry partners.
Universal Protection
Read more...
Innovative Production
Read more...
Clinical Trials
Read more...
The DeltaFLU Difference
DeltaFLU is the only vaccine in clinical development based on deletion of the influenza NS1 gene. Removing this influenza virulence gene gives DeltaFLU both its unique mode of attenuation and its highly effective mechanism of action. Lacking NS1, DeltaFLU rapidly induces interferon, a key component of the immune response to viral infection.
Deletion of NS1 achieves two highly advantageous properties of DeltaFLU:
Delivered into the nasal passages in a gentle spray, DeltaFLU introduces to the immune system natural viral antigens that stimulate multiple components of the immune system to achieve a broadly protective immune response. The effect is similar to infection with a circulating influenza strain, but with a crucial difference: DeltaFLU vaccine strains are replication-deficient. Because of this, DeltaFLU strains cannot cause disease and do not produce viral progeny. Individuals immunized with DeltaFLU do not shed the vaccine virus. These are important safety aspects of the DeltaFLU approach, and significant advantages over live vaccines that are able to replicate.
DeltaFLU vaccine strains can’t fight off the body’s interferon response because unlike naturally occurring influenza viruses, they lack the ability to produce NS1. As a result, DeltaFLU rapidly induces high levels of interferon and IgA antibodies in the linings of the nasal passages, creating a first line of defense against circulating disease-causing influenza viruses directly at their point of entry. This potent interferon response generates a self-adjuvant effect that activates a broadly protective systemic immune response in the form of serum antibodies, T cells, and T cell-based memory to viral infection. Prime-boost immunization with paired DeltaFLU vaccines also enables universal protection by focusing the immune response to conserved “stem” regions of the HA antigen shared by all influenza virus strain types.
The Need
Influenza is a serious public health threat. No infectious disease has higher rates of incidence and mortality. Worldwide, a third of all children and 10% of all adults are infected with influenza viruses every year. Influenza and its serious complications cause a half a million deaths annually.
data source: https://www.cdc.gov/flu/vaccines-work/past-seasons-estimates.html
Vaccination is the primary means of preventing influenza infections and their spread, but conventional influenza vaccines are only about 50% effective at best. Effectiveness of conventional vaccines relies on a good match between the strains on which the vaccines are based and strains circulating in populations. As influenza viruses replicate they undergo constant genetic change. The gradual accumulation of genetic changes results in “drifted” strains that can’t be recognized by antibodies generated by prior immunization or virus exposure. Conventional vaccine technologies require annual attempts to match influenza vaccines to the strains expected to dominate the coming flu season. Mismatches between vaccines and circulating strains frequently occur, resulting in poor protection.
Occasionally influenza viruses undergo an abrupt genetic change so significant that most people have no immunity to the new virus strain. Such “shifted” strains can cause an influenza pandemic. Rapid development, production and distribution of a new vaccine is paramount to protect the population and forestall the disease’s spread. Pandemic influenza is a relentless global threat. The 2009 influenza pandemic led to over a quarter of a million deaths. The three major influenza pandemics of the 20th century caused a combined total of over 50 million deaths.
DeltaFLU shows the potential to provide protection against all strains of influenza type A and B viruses, including drifted strains and emerging strains with pandemic potential. Clinical studies show DeltaFLU induces broadly cross-neutralizing antibodies in the nasal passages and serum that can block widely unmatched influenza strains. We have demonstrated the feasibility of universal protection with DeltaFLU in nonclinical models. These findings support our plans to evaluate universal protection with DeltaFLU in placebo-controlled studies in healthy volunteers. GMP production of DeltaFLU for these upcoming clinical studies is underway.